-
- Find Care
-
- Visitor Information
- Find a Location
- Shuttles
- Visitor Policies
-
-
- Our Virtual Care Options
- 24/7 Virtual Care
- Virtual Visits for Primary & Specialty Care
- Online Second Opinions
- Participate in Research
-
- Contact us
-
- For Innovators
- Commercialization Guide for Innovators
-
-
- Research News
- Alzheimer's Disease
- Artificial Intelligence
-
- Overview
-
- Overview
- Getting Started
- New to Mass General Brigham
- International Patient Services
- What Is Patient Gateway?
- Planning Your Visit
- Find a Doctor (opens link in new tab)
- Appointments
- Patient Resources
- Health & Wellness
- Flu, COVID-19, & RSV
- Billing & Insurance
- Financial Assistance
- Medicare and MassHealth ACOs
- Participate in Research
- Educational Resources
- Visitor Information
- Find a Location
- Shuttles
- Visitor Policies
- Find Care
-
- Overview
- Our Virtual Care Options
- 24/7 Virtual Care
- Virtual Visits for Primary & Specialty Care
- Online Second Opinions
-
- Overview
- Participate in Research
-
- Overview
- About Innovation
- About
- Team
- News
- For Industry
- Venture Capital and Investments
- World Medical Innovation Forum (opens link in new tab)
- Featured Licensing Opportunities
- For Innovators
- Commercialization Guide for Innovators
- Contact us
-
- Overview
- Information for Researchers
- Compliance Office
- Research Cores
- Clinical Trials
- Advisory Services
- Featured Research
- Two Centuries of Breakthroughs
- Advances in Motion (opens link in new tab)
- Brigham on a Mission (opens link in new tab)
- Gene and Cell Therapy Institute
- Research News
- Alzheimer's Disease
- Artificial Intelligence
-
- Overview
-
- Overview
- Residency & fellowship programs
- Brigham and Women's Hospital
- Massachusetts General Hospital
- Mass Eye and Ear
- Newton-Wellesley Hospital
- Salem Hospital
- Integrated Mass General Brigham Programs
- Centers of Expertise
- Global & Community Health
- Health Policy & Management
- Healthcare Quality & Patient Safey
- Medical Education
- For trainees
- Prospective trainees
- Incoming trainees
- Current trainees
- Continuing Professional Development
Preliminary Clinical Trial Results Show ‘Dramatic and Rapid’ Regression of Glioblastoma after Next Generation CAR-T Therapy
 Members of the Mass General Cancer Center INCIPIENT team (from left to right): Elizabeth Gerstner, MD, William Curry, MD, Marcela Maus, MD, PhD, Bryan Choi, MD, PhD, Kathleen Gallagher, PhD and Matthew Frigault, MD. Photo credit: Kate Flock/MGH.
Members of the Mass General Cancer Center INCIPIENT team (from left to right): Elizabeth Gerstner, MD, William Curry, MD, Marcela Maus, MD, PhD, Bryan Choi, MD, PhD, Kathleen Gallagher, PhD and Matthew Frigault, MD. Photo credit: Kate Flock/MGH.
Mass General Cancer Center researchers took a new approach to CAR-T, engineering CAR-TEAM cells to treat mixed cell populations within tumors
Working in collaboration with Mass General neurosurgeons, the team tested the approach in a phase 1 clinical trial of patients with recurrent glioblastoma
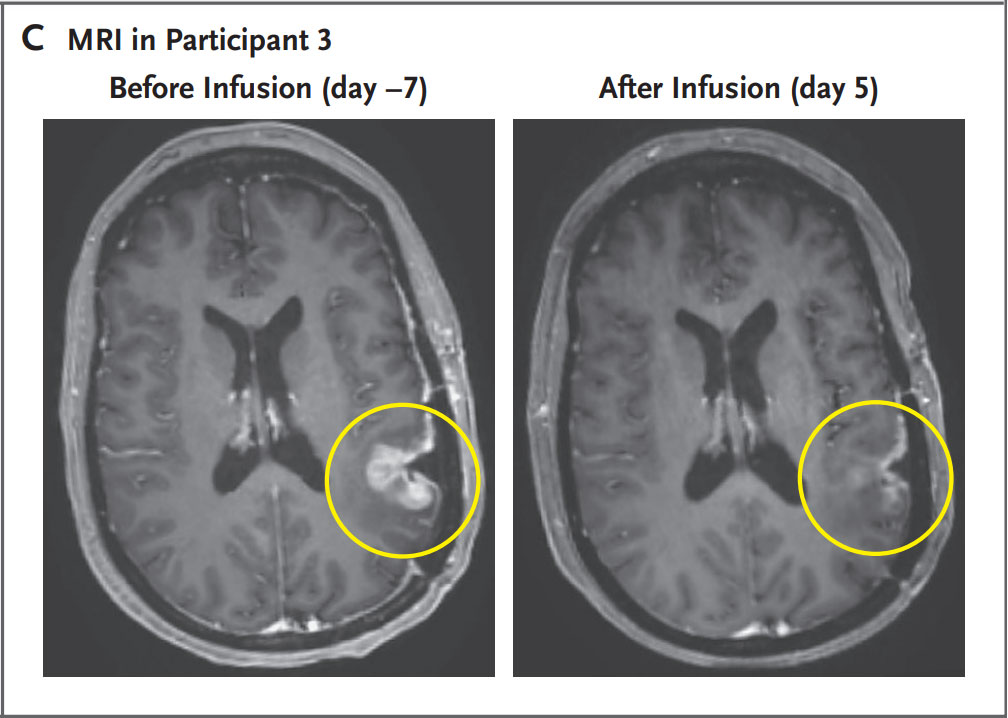
First three patients in the trial showed dramatic responses within days
A collaborative project to bring the promise of cell therapy to patients with a deadly form of brain cancer has shown dramatic results among the first patients to receive the novel treatment. In a paper published today in The New England Journal of Medicine, researchers from the Mass General Cancer Center, a member of the Mass General Brigham healthcare system, shared the results for the first three patient cases from a phase 1 clinical trial evaluating a new approach to CAR-T therapy for glioblastoma (GBM). The trial, known as INCIPIENT, is designed to evaluate the safety of CARv3-TEAM-E T cells in patients with recurrent GBM. Just days after a single treatment, patients experienced dramatic reductions in their tumors, with one patient achieving near-complete tumor regression. In time, the researchers observed tumor progression in these patients, but given the strategy’s promising preliminary results, the team will pursue strategies to extend the durability of response.
“This is a story of bench-to-bedside therapy, with a novel cell therapy designed in the laboratories of Massachusetts General Hospital and translated for patient use within five years, to meet an urgent need,” said Bryan Choi, MD, PhD, neurosurgeon and associate director of the Center for Brain Tumor Immunology and Immunotherapy, Cellular Immunotherapy Program, Mass General Cancer Center and Department of Neurosurgery. “The CAR-T platform has revolutionized how we think about treating patients with cancer, but solid tumors like glioblastoma have remained challenging to treat because not all cancer cells are exactly alike and cells within the tumor vary. Our approach combines two forms of therapy, allowing us to treat glioblastoma in a broader, potentially more effective way.”
The new approach is a result of years of collaboration and innovation springing from the lab of Marcela Maus, MD, PhD, director of the Cellular Immunotherapy Program at the Mass General Cancer Center, Paula O’Keeffe chair in Oncology, and faculty of the Krantz Family Center for Cancer Research. Maus’ lab has set up a team of collaborating scientists and expert personnel to rapidly bring next generation genetically modified T cells from the bench to clinical trials in patients with cancer.
“We’ve made an investment in developing the team to enable translation of our innovations in immunotherapy from our lab to the clinic, to transform care for patients with cancer,” said Maus. “These results are exciting, but they are also just the beginning—they tell us that we are on the right track in pursuing a therapy that has the potential to change the outlook for this intractable disease. We haven’t cured patients yet, but that is our audacious goal.”
Studies like this one show the promise of cell therapy for treating incurable conditions. Mass General Brigham’s Gene and Cell Therapy Institute, where Maus is Associate Head & Head of Cell Therapies, is helping to translate scientific discoveries made by researchers into first-in-human clinical trials and, ultimately, life-changing treatments for patients. The Institute’s multidisciplinary approach sets it apart from others in the space, helping researchers to rapidly advance new therapies and push the technological and clinical boundaries of this new frontier.
Media Contact
About Mass General Brigham
Mass General Brigham is an integrated academic health care system, uniting great minds to solve the hardest problems in medicine for our communities and the world. Mass General Brigham connects a full continuum of care across a system of academic medical centers, community and specialty hospitals, a health insurance plan, physician networks, community health centers, home care, and long-term care services. Mass General Brigham is a nonprofit organization committed to patient care, research, teaching, and service to the community. In addition, Mass General Brigham is one of the nation’s leading biomedical research organizations with several Harvard Medical School teaching hospitals. For more information, please visit massgeneralbrigham.org.